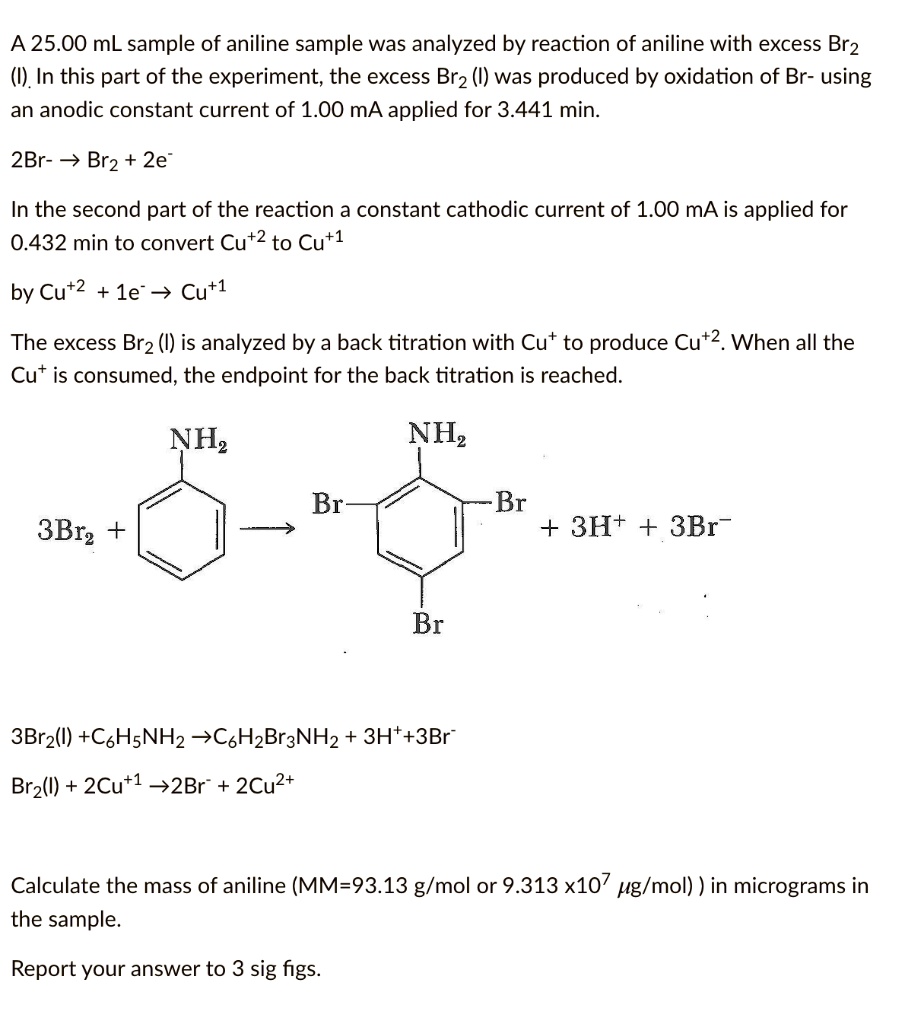
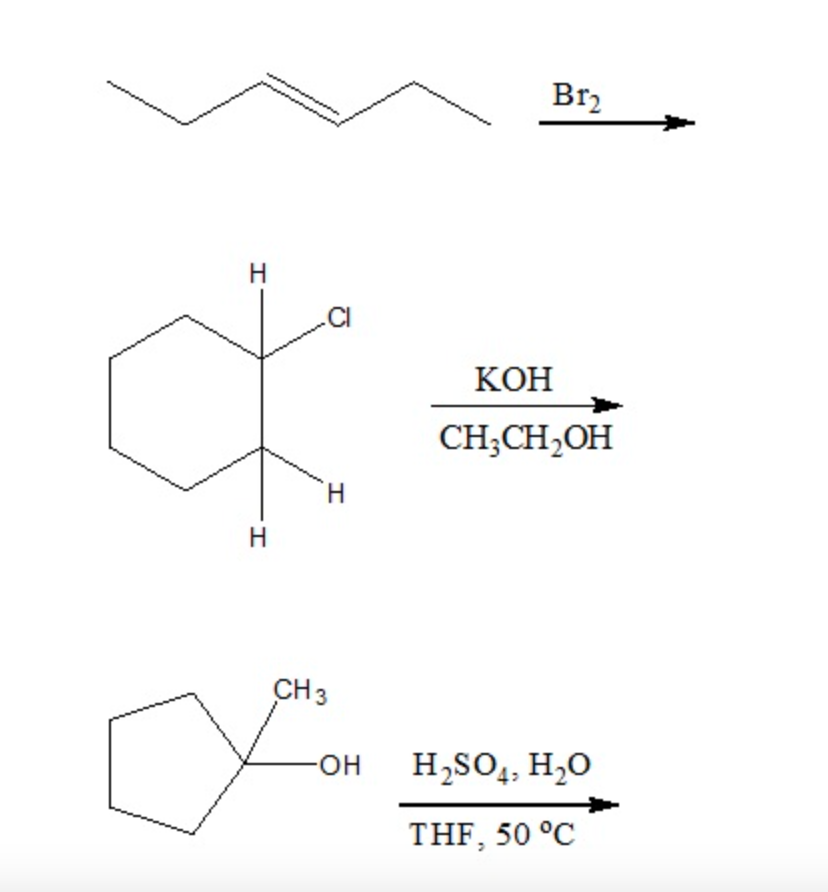
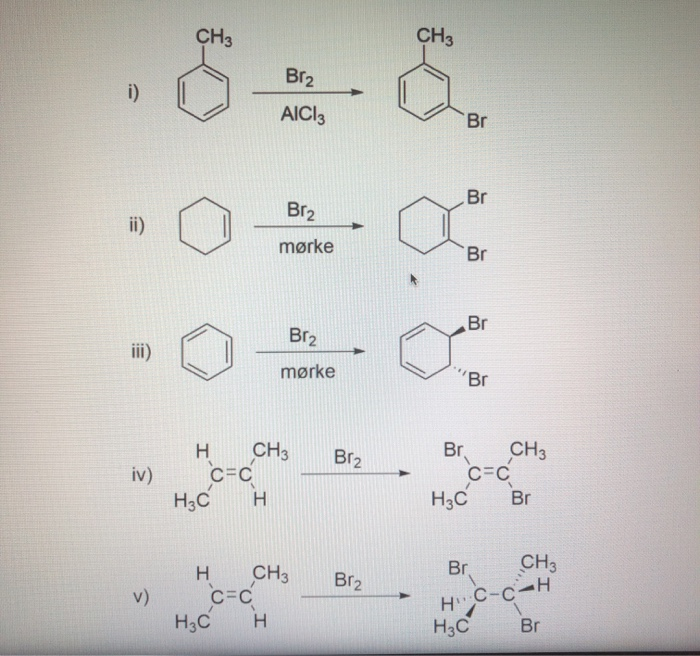
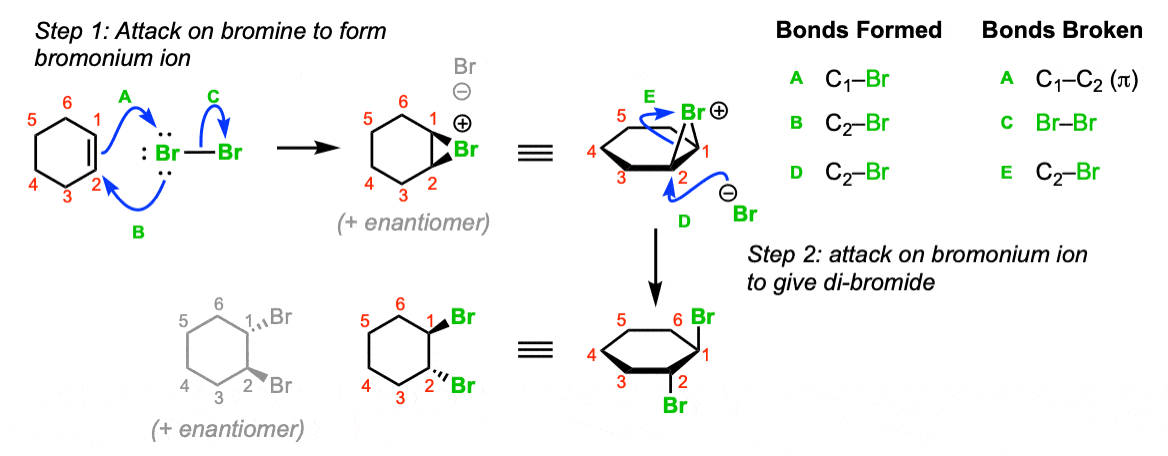
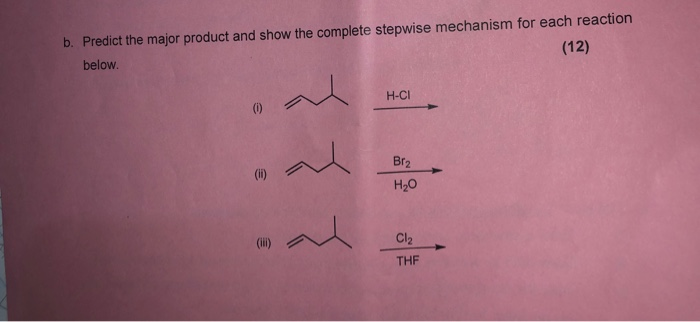
Bromine Radical (Br• and Br2•–) Reactivity with Dissolved Organic Matter and Brominated Organic Byproduct Formation | Environmental Science & Technology

⚗️Which of these statements are true? Select all that apply. The Delta.Hf for Br2(I) is 0 kJ/mol. The - Brainly.com

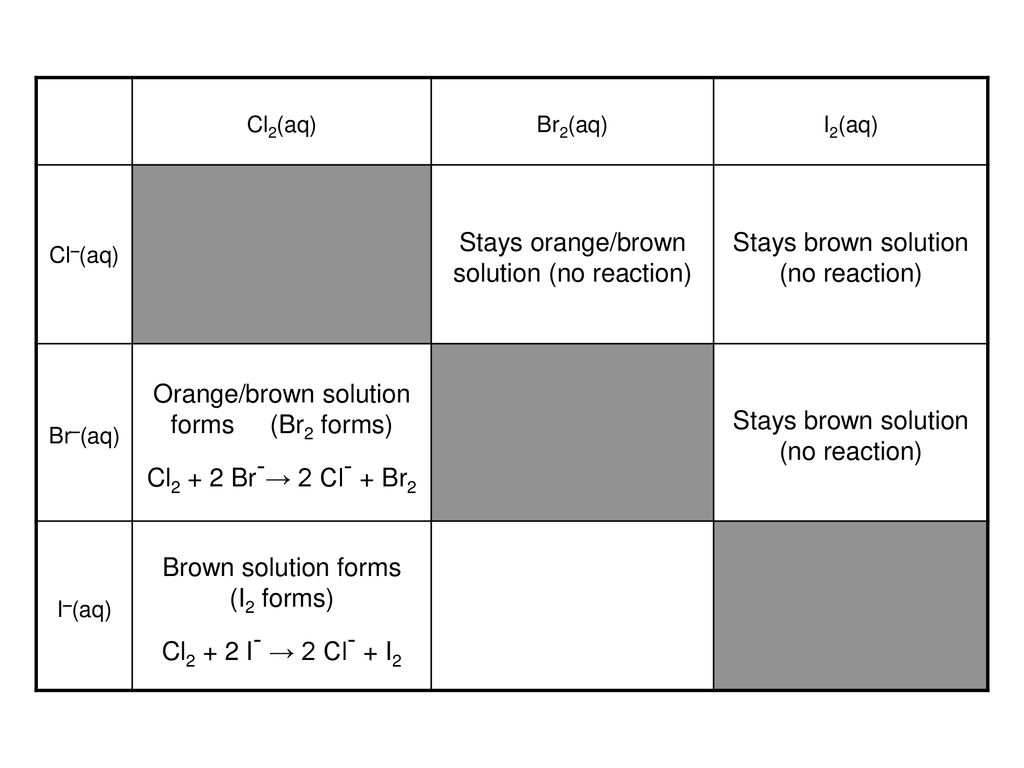
I2 and Br2 are added to a solution containing Br– and I– ions. What reaction will occur if, I(2) + 2e^(-) rarr 2I^(-), E^(0) = + 0.54V and Br(2) + 2e^(-) rarr 2Br^(-), E^(0) = +1.09 V?

SOLVED: Write a BALANCED equation for 4-methylcyclohexene with potassium permanaganate and a BALANCED equation for 4-methylcyclohexene with Bromine ( Br2). I am having trouble balancing the equation