Bisphenol A Analogs Induce Cellular Dysfunction in Human Trophoblast Cells in a Thyroid Hormone Receptor-Dependent Manner: In Silico and In Vitro Analyses | Environmental Science & Technology

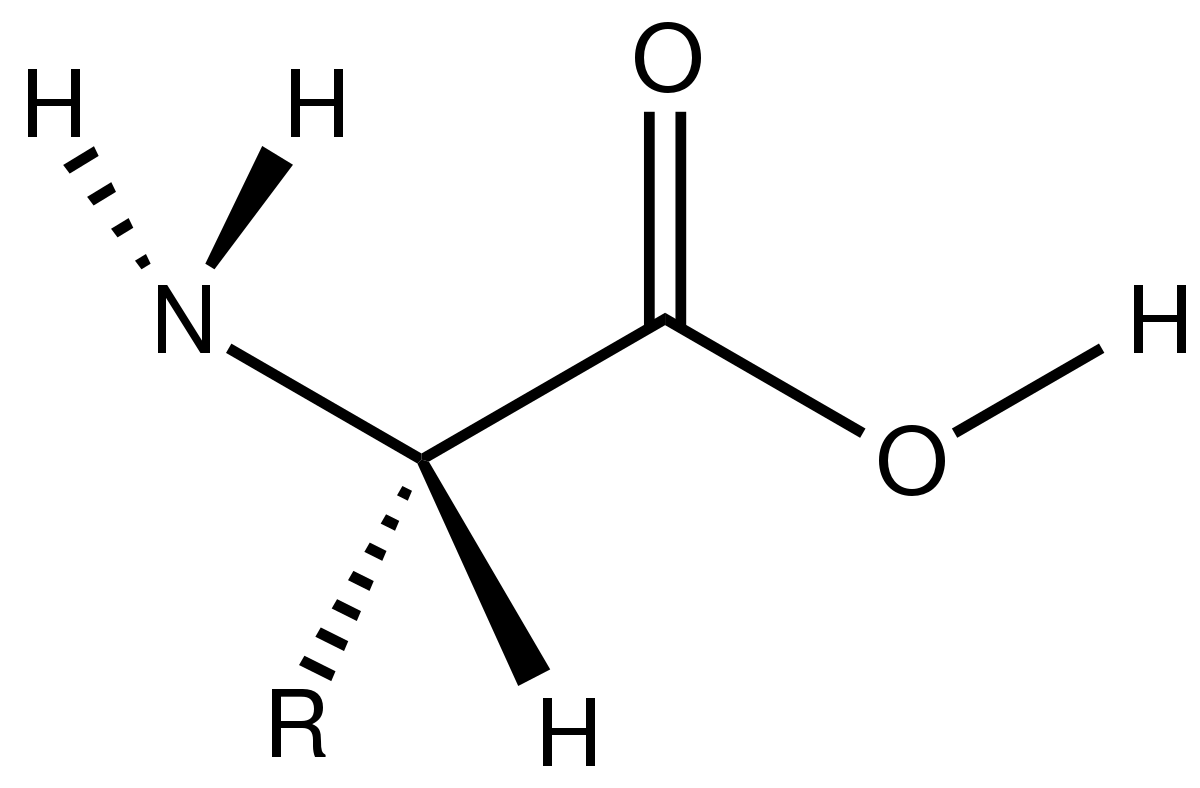
Advances in the Simulation of Protein Aggregation at the Atomistic Scale | The Journal of Physical Chemistry B

Amazon.com: Pro-line Racing 1/10 Trencher LP Front/Rear 2.8" MT Tires Mounted 12mm Blk Raid 2 PRO1015910 : Toys & Games
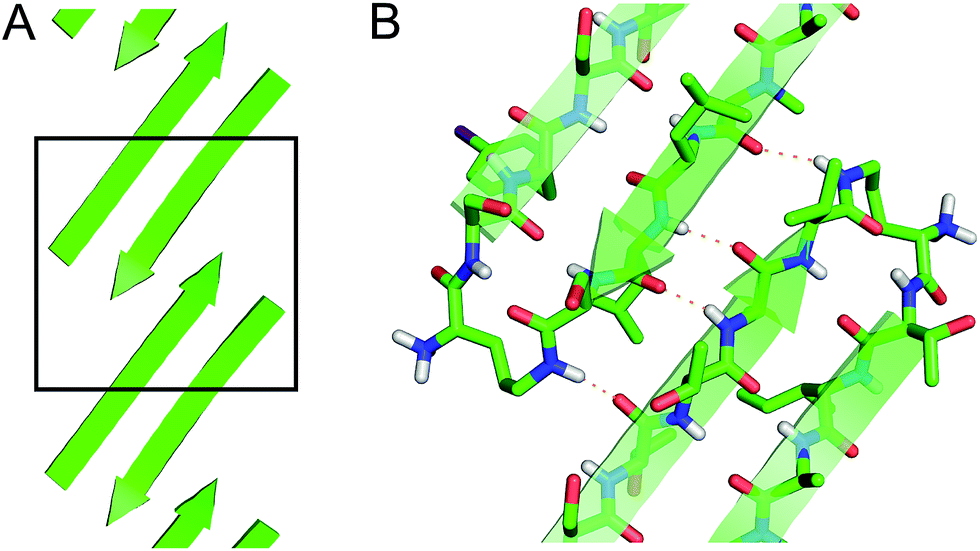
Square channels formed by a peptide derived from transthyretin - Chemical Science (RSC Publishing) DOI:10.1039/C6SC01927G
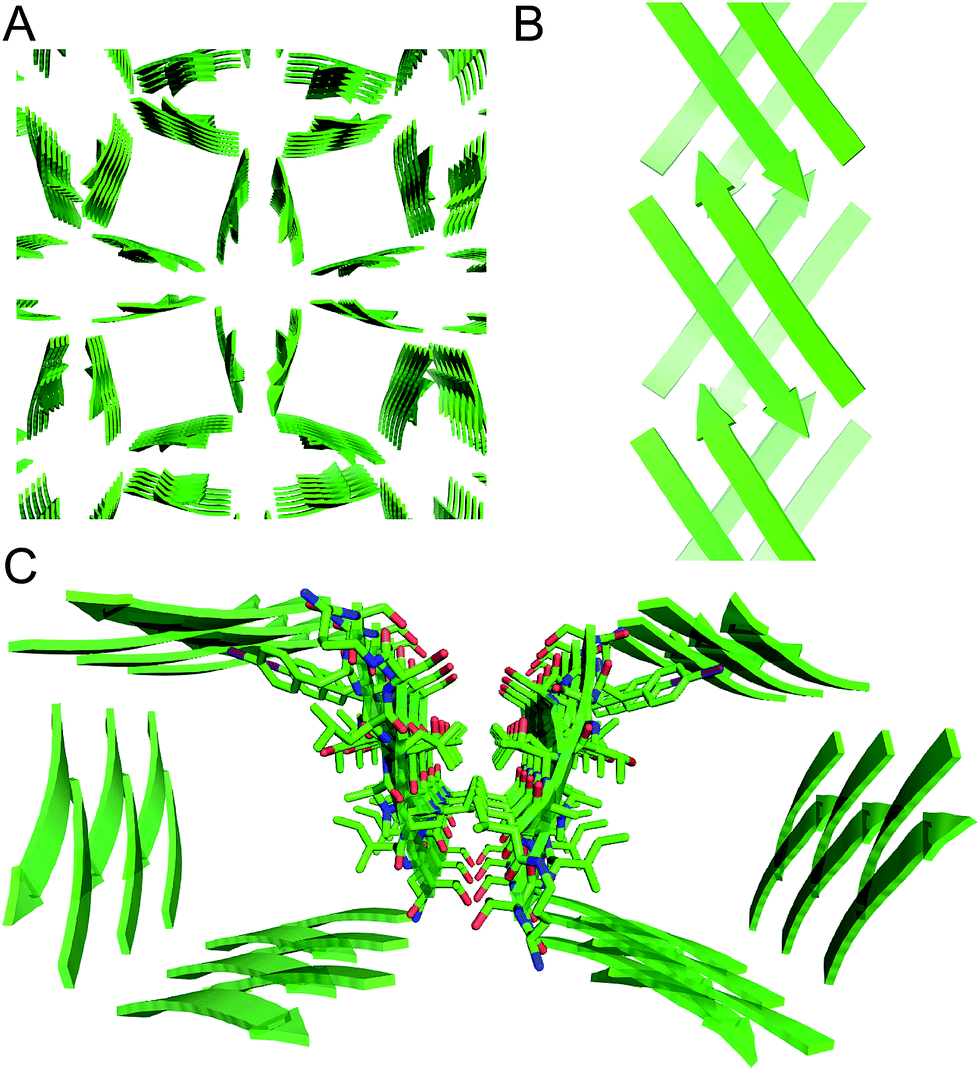
Square channels formed by a peptide derived from transthyretin - Chemical Science (RSC Publishing) DOI:10.1039/C6SC01927G
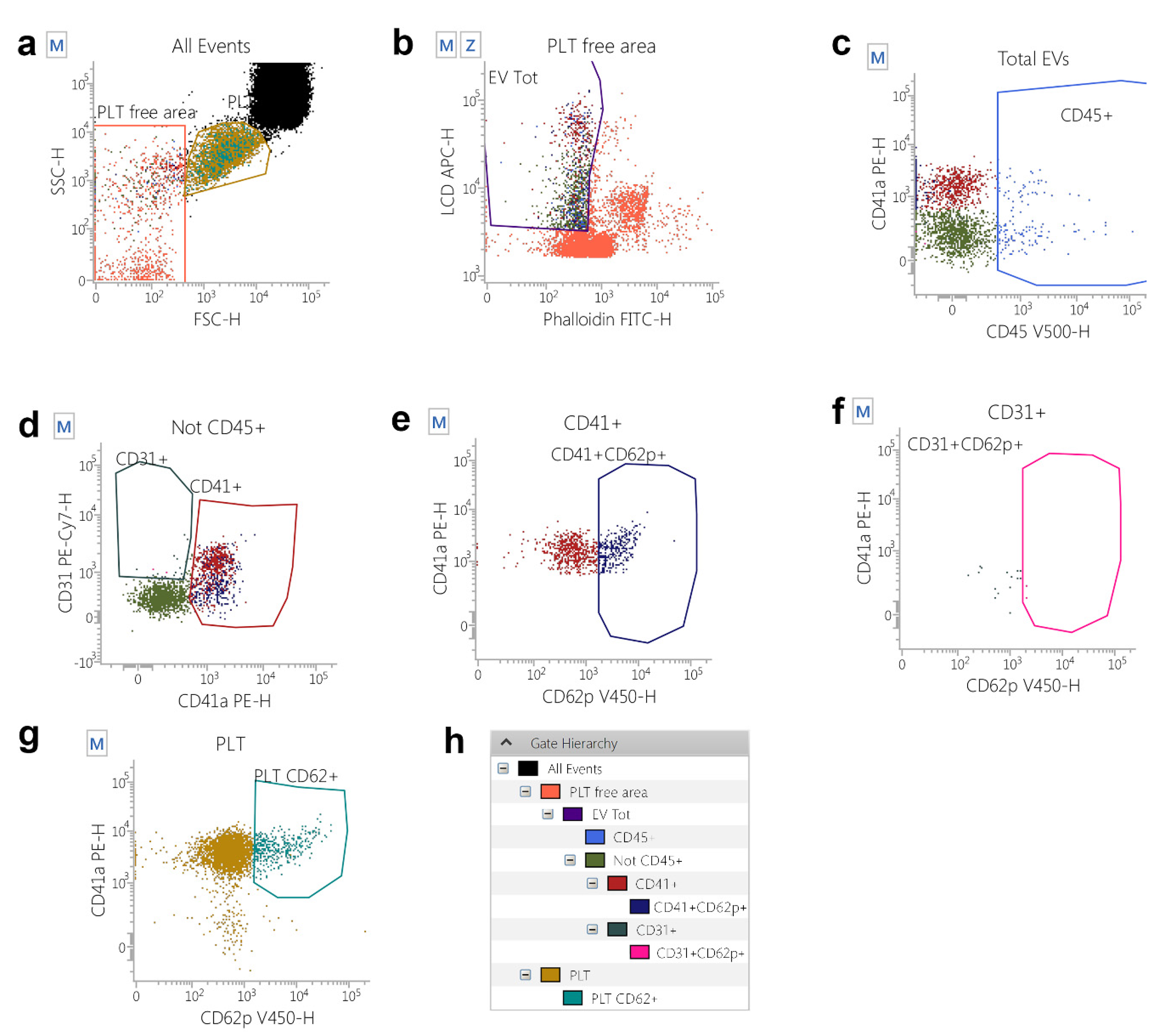
IJMS | Free Full-Text | Extracellular Vesicles in Regenerative Processes Associated with Muscle Injury Recovery of Professional Athletes Undergoing Sub Maximal Strength Rehabilitation
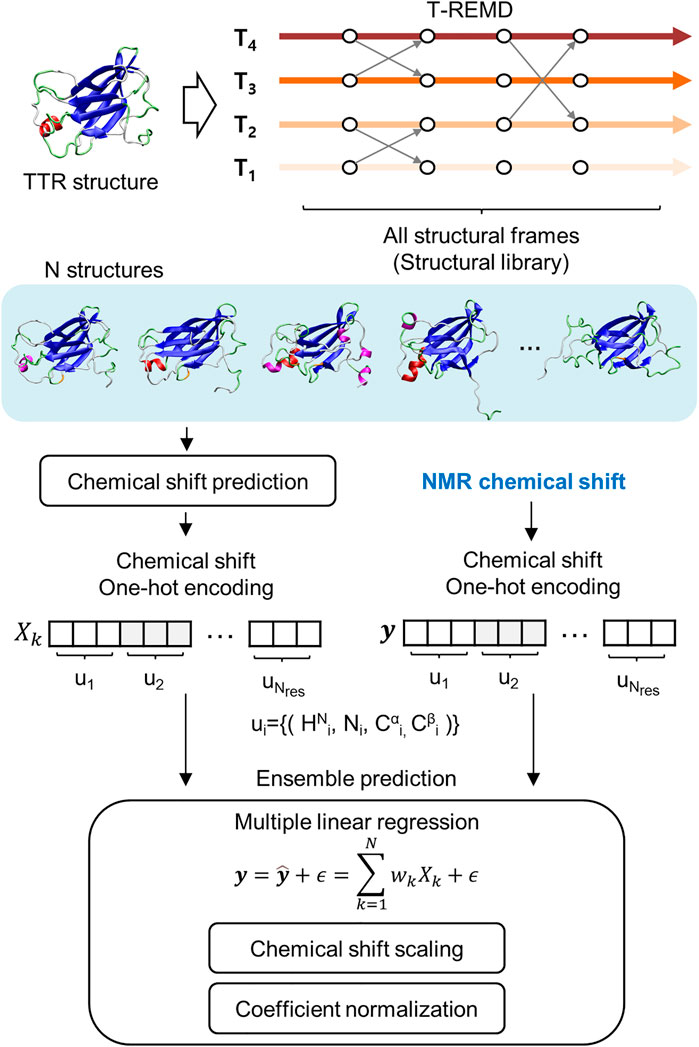
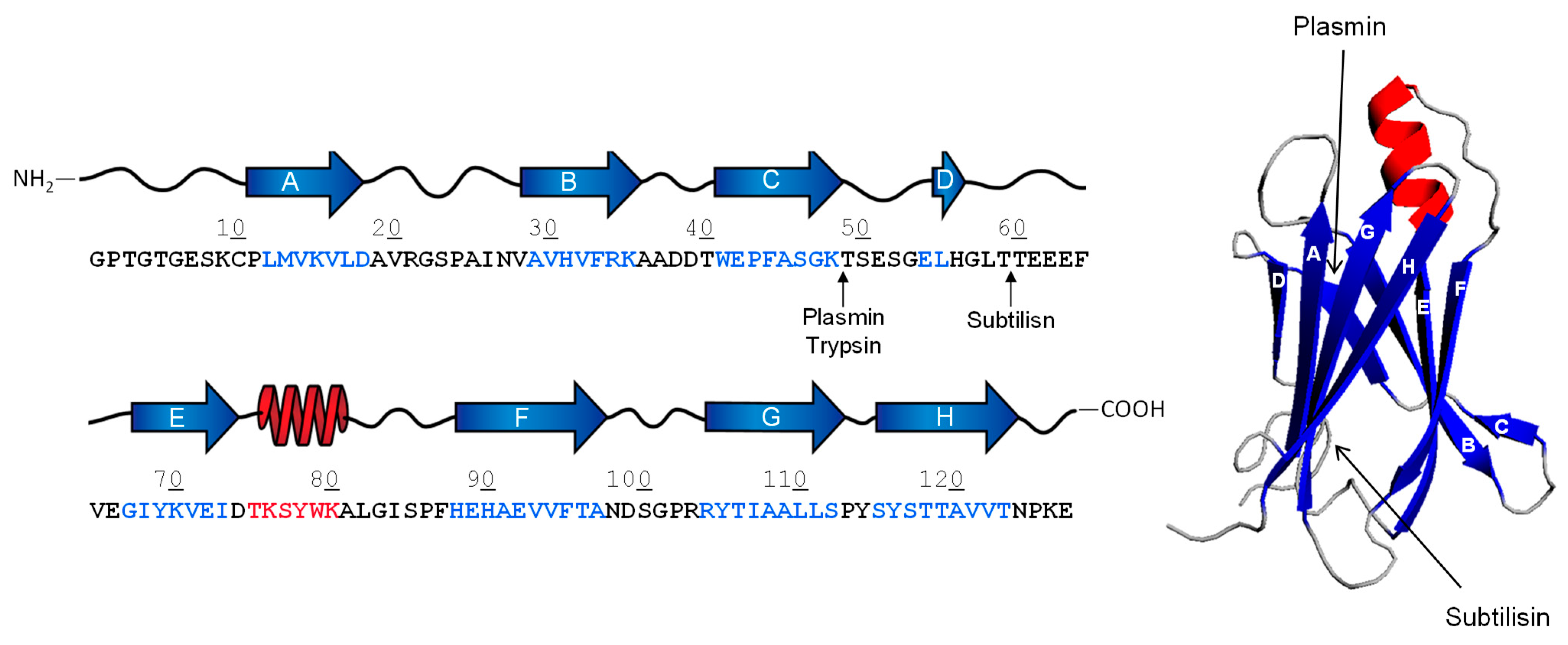
Frontiers | Aggregation-Prone Structural Ensembles of Transthyretin Collected With Regression Analysis for NMR Chemical Shift

Multi-eGO: an in-silico lens to look into protein aggregation kinetics at atomic resolution | bioRxiv
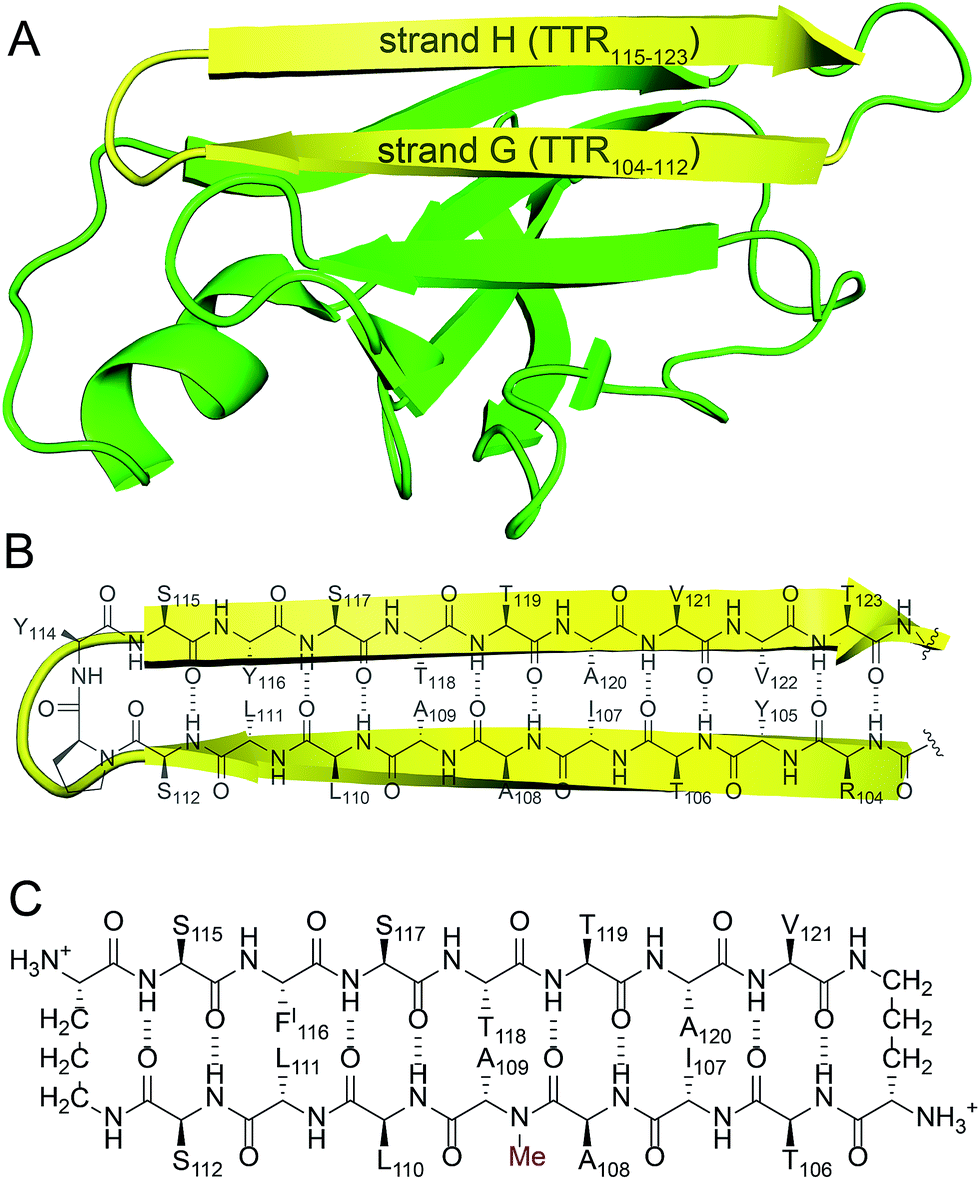
Square channels formed by a peptide derived from transthyretin - Chemical Science (RSC Publishing) DOI:10.1039/C6SC01927G

Multi-eGO: an in-silico lens to look into protein aggregation kinetics at atomic resolution | bioRxiv
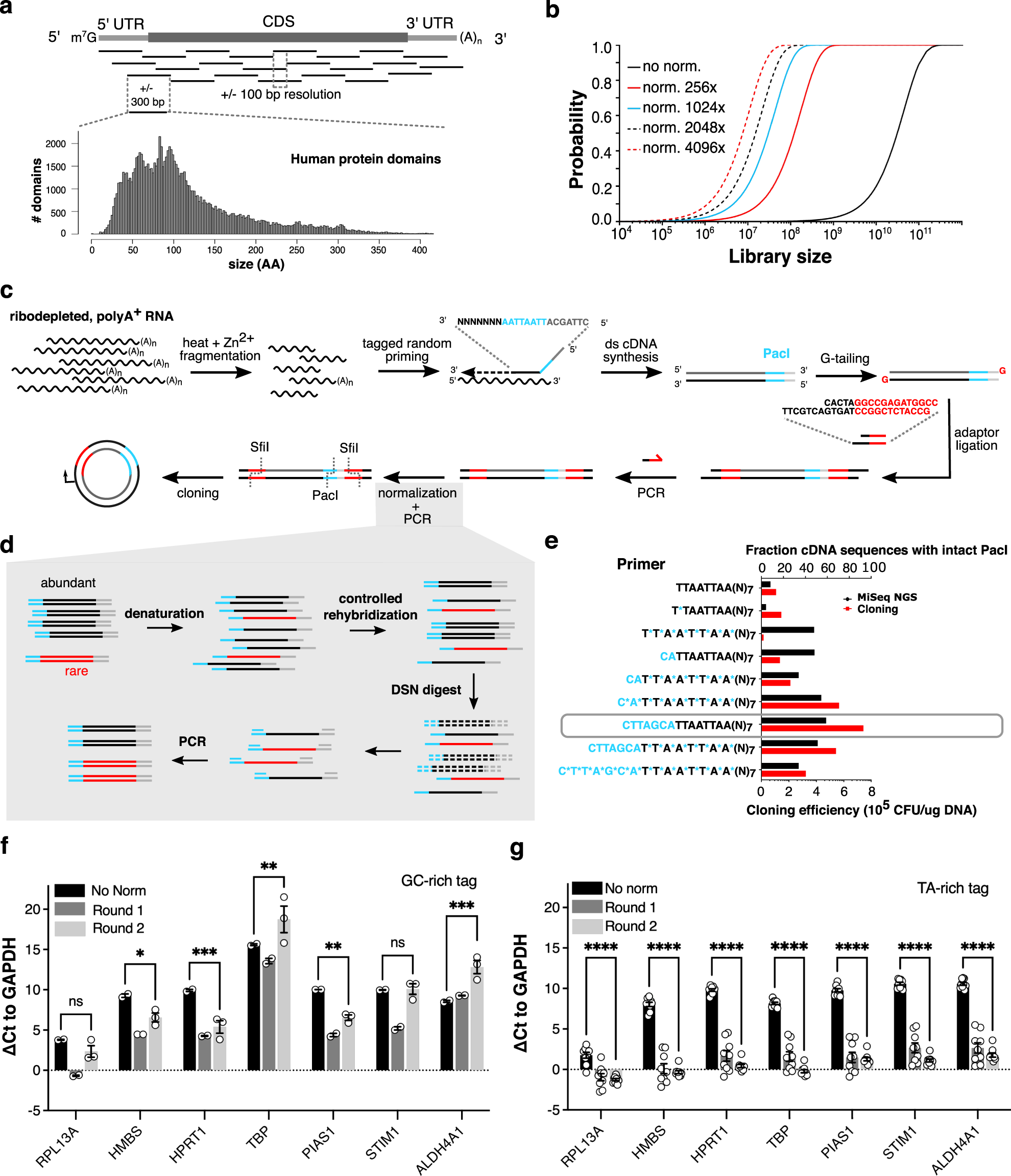
Massively parallel interrogation of protein fragment secretability using SECRiFY reveals features influencing secretory system transit | Nature Communications