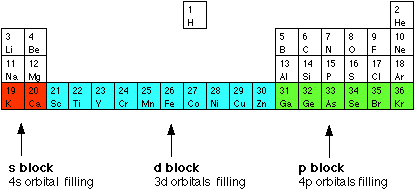
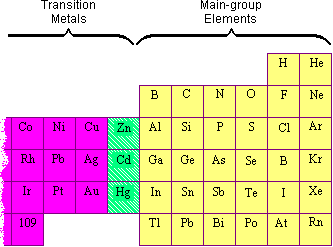
Why do the transition elements have higher enthalpies of atomization? In 3d series ( Sc to Zn ), which element has the lowest enthalpy of atomization and why?
Why are scandium and zinc not regarded as transition metals despite their been in the transition series? - Quora